  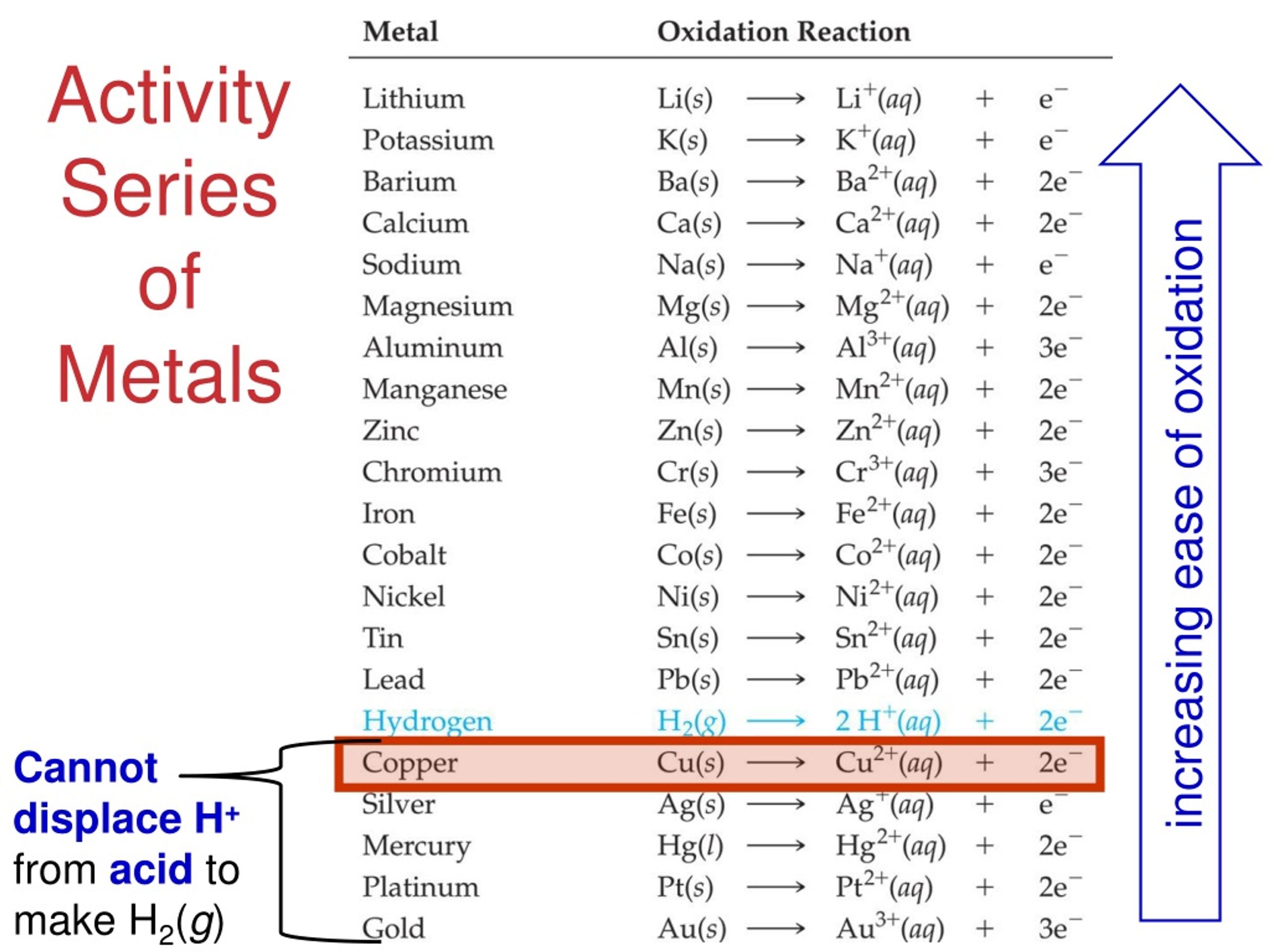
Hence, the upper part of the series contains powerful reducing agents as they are easily oxidized, and corrode easily. The reducing power of the metals decreases as we move from the top of the table to its bottom.The top of the reactivity series comprises the most reactive elements, which are not available in nature in the free state and the bottom of the series includes the least reactive elements.Here are the salient features of the reactivity series: Salient Features of the Reactivity Series However, metals from Zinc to Mercury can be easily extracted by reducing their oxides, which is comparatively cheaper. Electrolysis is used to extract metals that are placed higher in the reactivity series.On the other hand elements such as magnesium, iron, and aluminium react with steam to form oxides and hydrogen gas. Due to their high reactivity, Potassium, Sodium, Lithium, and, Calcium reacts with cold water to form hydroxides and hydrogen gas.But, different metals have different reactivities toward oxygen such as gold, which is an unreactive metal and doesn’t readily form oxides when exposed to air. Most of the metals react with oxygen to form metal oxides.Higher ranking metals generally show a more metallic nature than that placed lower in the series.The higher the metal placed in the series, the more reactive it is and the more strongly it reacts with water, oxygen, and acid.Reactivity series can be used to predict if a metal can displace another in a single displacement reaction.Knowledge of chemistry is endless, so to know more about various types of chemical reactions.Reactivity series of metals, also referred to as the activity series, is the arrangement of metals in the descending order of their activities. Therefore, nonmetals that gain electrons rapidly are more active than others. We determine the reactivity of nonmetals, upon their capability to gain electrons in the solution state to form negatively charged ions or anions. What determines the reactivity of nonmetals?Īns. Therefore, the more readily metal loses its electrons, the more active it is, and higher up it is in the reactivity series. We determine the reactivity of metals, upon their capability to lose electrons in the solution state to form positive ions or cations. What determines the reactivity of metals?Īns. Fluorine is the most active nonmetals and it displaces all other nonmetals in salt solutions. Which is the most active or reactive nonmetal?Īns. Due to its low reactivity, it found uses in the manufacture of laboratory equipment, electrodes, platinum resistance thermometers, dentistry equipment, and jewelry. It has remarkable resistance to corrosion, even at high temperatures, and is therefore considered a noble metal. Platinum is one of the least reactive metals and therefore lies at the bottom of the reactivity series. Which is the least active or reactive metal?Īns. The table below, explain the reactions of metals with oxygen (air), water, dilute acids, and other salt solutions, at various level of the reactivity series. Metals at the top of the reactivity series are difficult to obtain from their ores.The activity series facilitates the comparative study of the metals in terms of the degree of their reactivity.Metals at the top of the reactivity series have the ability to displace metals that are placed lower from their salt solutions.It is because, like metals, it too loses an electron and becomes a positively charged (H +) ion. Although a non-metal, hydrogen is included in the reactivity series of metals.The ease, with which a metal in solution loses electrons and forms a positive ion, decreases down the series, i.e., from cesium to platinum.Special Features of the Reactivity or Activity Series of Metals: Among the most commonly known metals, the most active cesium is at the top and the least active platinum is at the bottom of the reactivity series. The more readily metal loses its electrons, the more active it is, and the higher up it is in the reactivity series. The activity of a metal depends upon its capability to lose electrons in the solution state to form positive ions. For example, zinc being more active than copper replaces it with copper sulfate in the solution state to form zinc sulfate and free copper. In a displacement reaction, a metal higher up in the reactivity series displaces all other metals in a compound, which lies below it. It helps us to predict whether particular metal can displace another metal from a compound or not. Metal activity or reactivity series finds its utility in the study of displacement reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
